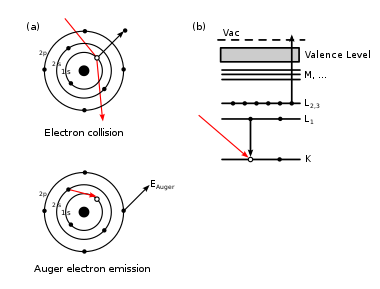
Auger effect ( Auger effect ) - electron emission of the atomic shell due to the nonradiative transition in the atom when excitation is removed, resulting from the formation of a vacancy for one reason on one of the inner shells. A vacancy may occur when another electron is knocked out by X-ray or gamma radiation, electron impact, or as a result of nuclear processes — internal conversion during the transition between nuclear levels or electron capture by a nucleus (one of the beta decay types) [1] . This phenomenon was first discovered and published in 1922 by Lisa Meitner [2] . Pierre Auger , whose name had the effect, independently discovered it in 1923 on the basis of an analysis of experiments in the Wilson chamber [3] .
The state of a positive ion with a vacancy formed on the inner electron shell is unstable, and the electron subsystem tends to minimize the excitation energy by filling the vacancy with an electron from one of the overlying electronic levels. The energy released during the transition to the underlying level can be either emitted in the form of a quantum of characteristic X-ray radiation , or transferred to the third electron, which is forced to leave the atom. The first process is more probable at an electron binding energy exceeding 1 keV , the second for light atoms and an electron binding energy not exceeding 1 keV .
The second process is called by the name of its discoverer, Pierre Auger , the “Auger Effect,” and the electron released, with the excess energy transferred to it, is Auger-electron . The kinetic energy of the Auger electron does not depend on the energy of the exciting radiation, but is determined by the structure of the energy levels of the atom. The spectrum of Auger electrons is discrete (in contrast to the continuous spectra of electrons produced by beta decay of nuclei). The binding energy E of an electron, to which the excitation energy E is transferred in the Auger process, must be less than E in . The kinetic energy of the Auger electron is equal to the difference between the excitation energy and the binding energy: E to = E in - E St. Typical kinetic energies of Auger electrons for different atoms and transitions range from tens of eV to several keV.
After the departure of the Auger electron, a vacancy remains in its place, so the shell is still in the excited state (the residual excitation energy is equal to the binding energy of the outgoing Auger electron). A vacancy, if it is not at the highest level, is filled with an electron from a higher shell, and the energy is carried away by the emission of a characteristic x-ray photon or a new Auger electron. This happens until vacancies move to the uppermost shell (in a free atom) or are filled with electrons from the valence band (when the atom is in a substance). The free atom as a result of the Auger transition, initiated by electron knocking out by external radiation or the effect of internal conversion, becomes at least a doubly charged positive ion (the first ionization is electron knocking out, the second is the Auger electron emergence). As a result of the Auger effect initiated by electron capture, a singly charged positive ion can be formed (since the charge of the atomic nucleus decreases by one as a result of the electron capture).
The vacancy energy can be transferred with a nonzero probability to any of the electrons from the overlying levels; therefore, the spectrum of Auger electrons usually consists of many lines. The average time τ from the onset of a vacancy to its filling is finite (and small), so the Auger lines have a finite width Δ E ≈ ħ / τ ~ 1 ... 10 eV , corresponding to the decay width Γ of a given atomic state.
Auger transitions in condensed matter can occur due to the filling of vacancies with electrons in the valence band, as a result of which the width of Auger lines increases, compared to transitions in single atoms. Auger transitions can also occur in free molecules. The molecular Auger spectrum is significantly more complex than the Auger spectra of single atoms.
Bones-Kronig Transition
A special case of the Auger effect, in which a vacancy is filled with an electron of the external sublevel of the same shell, is called the Koster – Kronig transition. In the case when the emitted electron belongs to the same shell, the effect is called the Koster – Kronig superjunction. The Koster-Kronig effect was named after the Dutch physicists Dirk Koster and Ralph Kronig who discovered it.
Application
It is used in Auger spectroscopy - a method based on the analysis of the energy distribution of electrons resulting from the Auger effect.
Notes
- ↑ IUPAC Gold Book internet edition: Auger Effect .
- ↑ L. Meitner. Über die Entstehung der β-Strahl-Spektren radioaktiver Substanzen (German) // Zeitschrift für Physik : magazin. - 1922. - Bd. 9 , Nr. 1 . - S. 131—144 . - DOI : 10.1007 / BF01326962 . - .
- Aug P. Auger: Sur les rayons β secondaires produits un gaz par des rayons X , CRAS 177 (1923) 169-171.
See also
- Auger recombination
- Electronic capture
Links
- IUPAC Gold Book internet edition: " Auger electron ".
- Meitner L. Über die Entstehung der β-Strahl-Spektren radioaktiver Substanzen (German) // Z. Physik. - 1922. - Bd. 9 , H. 1 . - S. 131—144 . - DOI : 10.1007 / BF01326962 . - .
- Auger P. Sur les rayons β secondaires produits dans un gaz par des rayons X (fr.) // Comptes rendus hebdomadaires des séances de l'Académie des sciences. - 1923. - Vol. 177 , livr. 3 - P. 169-171 .
- Duparc OH Pierre Auger - Lise Meitner: Comparative contributions to the Auger effect (Eng.) // International Journal of Materials Research (formerly Zeitschrift fuer Metallkunde). - 2009. - Vol. 100 , iss. 09 - P. 1162 . - DOI : 10.3139 / 146.110163 . .
- Burhop EHS The Auger Effect and Other Radiationless Transitions (eng.) . - Cambridge Monographs on Physics, 1952.
- Chattarji D. The Theory of Auger Transitions (English) . - London: Academic Press, 1976 ..
Literature
- Parilis E.S. Auger Effect. - Tash. : Fan, 1969. - 211 p.