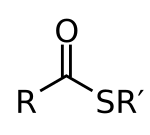
Thioesters are organic compounds containing the functional group CS-CO-C and which are thiol and carboxylic acid esters . Thioesters play an important role in biochemical processes, the most well-known representative of this class is acetyl-CoA .
Content
Getting
Methods for producing thioesters are diverse, [1] but the most important is the condensation of thiols and carboxylic acids in the presence of water-removing reagents [2] (for example, N , N ' - dicyclohexylcarbodiimide [3] ( DCC , DCC):
- RSH + R'CO 2 H → RSC (O) R '+ H 2 O
Carboxylic acid anhydrides and some lactones react in the same way with thiols in the presence of bases.
Thioesters are conveniently prepared by the Mitsunobu reaction from alcohols and thioacids (for example, thioacetates from thioacetic acid [4] ).
They can also be obtained by carbonylation of alkenes and alkynes in the presence of thiols. [five]
Reactions
The carbonyl group of thioesters is reactive with respect to nucleophile attacks (to a lesser extent than with acid chlorides ). So, complex thioesters react with amines to form amides :
Fukuyama's reaction is unique for thioesters: when interacting with an organozinc compound in the presence of a palladium catalyst, ketones are formed.
Hydrogen atoms in the α-position to the carbonyl group of thioesters exhibit moderate acidity; therefore, the thioesters are subject to aldol condensation . This reaction is important for the biosynthesis of fatty acids.
Biochemistry
Thioesters are common intermediates in many biochemical transformations, including the formation and breakdown of fatty acids and mevalonate , a synthetic precursor of steroids. Biosynthesis of lignins , which constitute a significant part of the biomass, proceeds through the formation of caffeic acid thioester. [6] Other examples of thioesters are malonyl-CoA, acetoacetyl-CoA, propionyl-CoA and cinnamoyl-CoA. Acetogenesis proceeds through the formation of acetyl-CoA . Thioesters are formed in living organisms as a result of esterification reactions, with ATP playing the role of a dehydrating agent. They also play a key role in the ubiquitination of proteins by ubiquitin ligases , which mark proteins for subsequent destruction by proteasomes .
It is assumed that the oxidation of the sulfur atom of thioesters (thiolactones) is a necessary step to activate a number of platelet aggregation inhibitors such as ticlopidine , clopidogrel and prasugrel . [7] [8]
Thionic esters
Thionic esters ( O -thiocarboxylic esters) are isomers of thioesters. The sulfur atom in the thionic esters replaces the carbonyl oxygen atom of the ester group, for example: C 6 H 5 C (S) OCH 3 (methylthionobenzoate). These compounds are usually obtained by the reaction of thioacyl chlorides with alcohols; they can also be obtained by treating esters with Lawson's reagent . [9]
Notes
- ↑ Fujiwara, S .; Kambe, N. Thio-, Seleno-, and Telluro-Carboxylic Acid Esters // Topics in Current Chemistry. - Berlin / Heidelberg: Springer, 2005. - Vol. 251. - p. 87–140. - ISBN 978-3-540-23012-0 . - DOI : 10.1007 / b101007 .
- ↑ Synthesis of thioesters Neopr . Organic Chemistry Portal. Archived October 29, 2012.
- ↑ Mori, Y .; Seki, M. (2007), " Synthesis of Multifunctionalized Ketones Through the Fukuyama Coupling Reaction: Catalyst Preparation of Ethyl 6-oxotridecanoate ", Org. Synth. T. 84: 285 , < http://www.orgsyn.org/orgsyn/orgsyn/prepContent.asp?prep=V84P0285 > ; Coll. Vol. T. 11: 281
- ↑ Volante, R. A new, highly efficient method for the conversion of alcohols to thiolesters and thiols (English) // Tetrahedron Letters : journal. - 1981. - Vol. 22 , no. 33 . - P. 3119-3122 . - DOI : 10.1016 / S0040-4039 (01) 81842-6 .
- ↑ Bertleff, W .; Roeper, M .; Sava, X. Carbonylation // Ullmann's Encyclopedia of Industrial Chemistry. - Weinheim: Wiley-VCH, 2005. - DOI : 10.1002 / 14356007.a05_217.pub2 .
- ↑ Lehninger, AL; Nelson, DL; Cox, MM Principles of Biochemistry. - 3rd. - New York: Worth Publishing, 2000. - ISBN 1-57259-153-6 .
- ↑ Mansuy, D .; Dansette, PM Sulfenic acids as reactive intermediates in xenobiotic metabolism (Eng.) // Archives of Biochemistry and Biophysics : journal. - Elsevier , 2011. - Vol. 507 , no. 1 . - P. 174-185 . - DOI : 10.1016 / j.abb.2010.09.015 .
- ↑ Dansette, PM; Rosi, J .; Debernardi, J .; Bertho, G .; Mansuy, D. Metabolic Activation of Prasugrel: Nature of the Two Competitive Pathways (Eng.) // Chemical Research in Toxicology : journal. - 2012. - Vol. 25 , no. 5 - P. 1058-1065 . - DOI : 10.1021 / tx3000279 .
- ↑ Cremlyn, RJ An Introduction to Organosulfur Chemistry. - Chichester: John Wiley and Sons, 1996. - ISBN 0-471-95512-4 .