The Heck reaction is a combination of alkyl and aryl halides catalyzed by palladium complexes with alkenes containing at least one hydrogen atom in a double bond . The reaction was discovered by the American chemist Richard Heck in 1968. The mistakenly used name “Mizoroki – Heck reaction” arose because of the numerous references to the 1971 work under the authorship of the Japanese chemist T. Mizoroki, where he also describes this reaction, and Heck’s work dated 1972, although he was the discoverer four years earlier [ 1] . Initially, the reaction was discovered as a combination of aryl mercury compounds with alkenes in the presence of palladium salts, but in the mid-1980s a catalytic variant and convenient methods for carrying out this reaction appeared [2] .
At present, the reaction is formally a replacement of a hydrogen atom with a double bond with an alkyl or aryl residue in the halide with the double bond configuration reversed [3] .
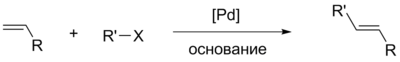
Content
Mechanism
At the first stage of the reaction, the oxidative addition of the halide to the unsaturated palladium atom occurs with the formation of an alkyl (aryl) palladium intermediate . Then coordination and introduction of the alkene molecule via the C-Pd bond occurs. The degree of oxidation of palladium increases from 0 to +2. Finally, at the third stage, β-elimination occurs with the formation of the reaction product, after which the catalyst regenerates under the action of the base [4] .
Catalyst Generation
The active catalytic particle, the diphosphine complex of Pd (PPh 3 ) 2, is formed from tetrakis (triphenylphosphine) palladium Pd (PPh 3 ) 4 as a result of the dissociation of two ligands [4] . Sometimes a catalyst is generated by the reduction of some Pd (II) complex: usually such a reduction occurs under the action of the phosphine ligand itself and the promotion of nucleophiles present in the mixture (hydroxides, alkoxides, water, or others). In the absence of phosphine, the role of a reducing agent is played by the amine, if it is used as a base, or by an alkene, a reducing catalyst according to the mechanism of the Wacker process . The problem at this stage is an insufficiently high concentration of the diphosphine complex in the reaction mixture, which decreases with an increase in the concentration of free ligand, as well as its tendency to disproportionate to the triphosphine ligand and unstable low-coordinated complexes that quickly aggregate into inactive metal particles [5] .
Oxidative addition
Oxidative addition is a typical process in organometallic chemistry in this case obeys general dependencies. The speed of this stage depends mainly on the nature of the group X and increases in the next row [4] :
In most cases, as a result of the addition of a halide to the complex, an evolved trans- product is formed, in which the groups R 'and X are located opposite each other. However, it was shown that the formation of a cis- product actually occurs, which then undergoes cis / trans- isomerization. Moreover, it is the cis complex that participates in the next stage [6] . In many cases, this stage determines the speed of the whole process [7] .
Implementation
To coordinate an alkene with a palladium complex, it is necessary that one of the ligands leave the coordination sphere of the metal. Two mechanisms have been proposed for this process, which differ depending on whether the charged or uncharged ligand leaves the complex: in the neutral mechanism, phosphine does this, as a rule, and in the cationic or charged mechanism, the complex leaves the anion X - . The choice of path depends on the nature of the ligands present in the complex. For monodentate phosphine ligands, the reaction is likely to proceed both along the neutral and cationic paths [6] .
At the implementation stage, the Heck regioselectivity is specified, which is determined by spatial and electronic factors. For a neutral and cationic type of reaction involving alkenes enriched with electrons, electronic factors are decisive: palladium is attached to a carbon atom having a higher electron density. Electronic factors easily overlap with spatial: a new CC bond is created at the less substituted carbon atom of the double bond. It is this criterion that is decisive in the intramolecular Heck reaction [8] .
β-elimination
At the β-elimination stage, the product is removed from the reaction sphere and palladium hydride complex is formed. This stage takes place after the conformation of the complex is changed to a more stable one, and the product of the E- configuration is formed. The process proceeds as syn- elimination (that is, palladium and hydrogen atoms split off on one side of the substrate molecule) [9] . The reversibility of the β-elimination step leads to the partial isomerization of the alkene. It can be avoided by using a silver salt as a base [10] [1] .
Reaction Conditions
Substrates
Diazonium salts
Typical substrates combined with alkenes in the Heck reaction are halides as well as triflates. In addition to them, other electrophiles have also been found that join the combination and allow it to be performed under milder conditions. As a first alternative, diazonium salts , in particular tetrafluoroborates, were tested. The reaction with diazonium salts occurs in the absence of base and phosphines, the addition of which leads to the decomposition of salt. In this case, apparently, the role of the base is performed by the alcohol used as a solvent. The use of diazonium salts accelerates the reaction, but also requires the use of a larger amount of catalyst (at least 1–2%), the reduction of which occurs due to the decomposition of the diazonium salt. In this case, the salt does not necessarily enter into the reaction in pure form: it can be obtained by the reaction of diazotization simultaneously with the Heck reaction. The greater activity of the diazo group compared to iodide was used in the stepwise assembly of complex molecules [11] .
Iodonium salts
Similar reagents are iodonium salts exhibiting reactivity similar to diazonium salts, but having greater resistance to bases. The high reaction rate and low temperatures make it possible to use this modification in difficult situations when, due to harsh conditions, it is impossible to apply the classic Heck reaction. At the same time, iodonium salts are not so accessible, therefore, their use in simple cases is unjustified [12] .
Iodonium salts contain two aryl or vinyl substituents at the iodine atom, and under normal conditions only one substituent is transferred to the alkene, whereas at 100 ° C both groups participate in the reaction. In order to avoid the loss of the sometimes valuable substituent, iodonium salts were proposed with one “unnecessary” substituent, the role of which is usually performed by the phenyl group . If a vinyl and aryl group is present in an asymmetric salt, then a vinyl group is involved in the reaction. Of the two aryl groups, the one that contains electron-donating groups is more active (despite the fact that the dependence is usually inverse to the oxidative addition of aryl halides). The reactions of cyclic iodonium salts are also described [12] .
Chlorides and anhydrides
Chlorides were suggested as alternative substrates rather early. The reaction with their participation includes a decarbonylation stage, after which it proceeds in accordance with the catalytic cycle of the Heck reaction. Decarbonylation is also the most demanding stage of the reaction, since for its occurrence three free coordination sites must be present in the complex, and any ligand with good binding inhibits the reaction. Therefore, it is better to conduct the reaction in non-polar solvents, in the presence of bulk amines incapable of coordination, and in the absence of phosphines. A similar reaction with the participation of anhydrides is remarkable in that it does not require the addition of a base, since the carboxylate ion released in the reaction can itself act as a base. This phenomenon is especially valuable in those cases when it is required to maximally simplify the composition of the reaction mixture [13] .
Catalysts
Typical conditions of the Heck reaction include the use of 1-5 mol. % palladium catalyst together with phosphine ligands and a suitable base (soluble as triethylamine , or insoluble as potassium carbonate, silver carbonate ). In the case of active halides (iodides), the required concentration of the catalyst is extremely low and can be reduced to parts per million . Triarylphosphines, N- acyl-substituted dipyridyls, N- acyl-substituted dipyrimidylamines, Pfaltz ligand and ligands based on ketopic acid can act as ligands [1] . Bidentate phosphines were considered bad ligands for the Heck reaction, but they did well in the cationic mechanism and began to be used in the enantioselective reaction variant, where precise control of the coordination sphere of the complex is important. In this case, instead of chlorides, it is necessary to use trifluoromethanesulfonates (triflates) and add silver or thallium salts to the reaction mixture, which does not add attractiveness to the method. Nevertheless, chelating phosphine ligands have positive sides: there is no need to use their excess to obtain a stable catalytic complex, the complexes themselves are more stable during storage, they have a large number of revolutions and can be reused [14] .
Herrmann's discovery of the catalytic activity of the Pd 2 (P ( o -Tol) 3 ) 2 (μ-OAc) 2 complex was a significant milestone in the development of the Heck reaction. This catalyst was named after him and was given a short designation of hc . It is one of the most convenient forms of palladium catalyst, easily synthesized by the reaction of palladium acetate and the corresponding phosphine, resistant to air and moisture. Its main advantage lies in the fact that it is an analogue of the palladium monophosphine complex, which is extremely difficult to obtain from both the multi-phosphine ligands and the mixture of the precursor with phosphine. At the same time, monophosphine complexes are very active and at the same time more stable than besfosphine complexes. Palladacycle Herrman is the most studied catalyst of this type. He very actively catalyzes combinations of aryl bromides ( sodium acetate , dimethylformamide, 100-140 ° C), especially those containing acceptor groups. In these cases, the speed of the catalyst reaches 10 6 [15] .
Another type of palladium catalysts for the Heck reaction are complexes with N- heterocyclic carbenes [16] .
A separate task is to create catalysts that can be easily regenerated and reused after the reaction. In the case of Heck's reaction, this task is rather difficult, since it involves many components, the composition of the mixture is constantly changing, the catalytic system is unstable and can be poisoned by salts formed as a result of the reaction of the base with evolved acid equivalent. The number of revolutions of the catalysts as a function of the reaction time drops sharply after a short period of activity. Nevertheless, work on the search for regenerable catalysts is underway, since the Heck reaction is of considerable industrial interest. The main idea of such developments is the creation of heterogeneous systems in which products and catalysts will be in different phases. Two-phase systems with two liquid phases and with liquid and solid phases have been developed. In the latter approach, the palladium catalyst was immobilized on polymers containing phosphine ligands and various inorganic substrates ( silica gel , zeolites , etc.) [17] .
Cheaper analogs can also be used as catalysts. Great prospects are associated with nickel . In particular, it has been shown that nickel can catalyze the Heck reaction using a stoichiometric amount of a reducing agent (for example, zinc dust). Obviously, the nickel catalyst is not regenerated as effectively as palladium. However, nickel has one serious advantage: it allows the use of aliphatic halides in the reaction. Examples of the use of salts of copper , platinum , rhodium , iridium and ruthenium are also described [18] .
Phase carriers
The role of phase transfer carrier is performed by quaternary ammonium salts. They can be carriers between the solid and liquid phases if bases that are practically insoluble in the typical Heck reaction solvents (acetates, carbonates, bicarbonates, sodium or potassium phosphates) are used in the reaction. They can also transfer between liquid phases if the base is soluble in water and the substrate is not. Halides, acetate and other ions accelerate the reaction, increasing the charge density on the palladium atom and facilitating the oxidative addition stage (this applies not only to ammonium salts, but also to metal salts). Quaternary ammonium salts can also stabilize low-coordinated palladium complexes or undergo ion exchange , for example, to replace iodine with chlorine in the complex in aryl iodide reactions. By such a replacement of the halogen ligand, a “switch” from the neutral to the charged mechanism occurs [19] .
It is also known that the use of Quaternary ammonium salts allows you to speed up the reaction and lower the temperature of its implementation. Thus, the reactions of aryl iodides can be carried out at temperatures below 100 ° C and even at room temperature. For this reason, the addition of a promoter salt has become a popular technique during the Heck reaction [19] .
Solvents
Many solvents are suitable for the Heck reaction, but dipolar aprotic solvents ( dimethylformamide , N- methyl pyrrolidone , dimethyl sulfoxide and acetonitrile ) are most often used because they have the ability to stabilize palladium complexes due to poor coordination. It also turned out that water can be used for the reaction. It can act as a ligand in a palladium complex, displacing other, less strongly bound ligands, for example, iodide, which poisons the catalyst and blocks the charged reaction mechanism. However, water can displace useful ligands, for example, acetate or bicarbonate. Water contributes to the creation of more compact transition states due to hydrophobic interactions. This affects the reaction rate and its selectivity, especially in the case of the intramolecular Heck reaction [20] .
As a medium in the Heck reaction, ionic liquids , which are liquid salts at room temperature, or molten salts can be used. They have high polarity and direct the reaction by a charged mechanism, and also stabilize low-coordinated palladium complexes. The simplest and recommended example of such a solvent is tetrabutylammonium bromide [21] .
In addition to halogenides, arylsilanes, stannans, triflates , bismuth and antimony compounds, diazonium salts, hypervalent iodine compounds, etc. can enter into a combination reaction with alkenes. [7] [1]
Industrial Value
As of 2001, the Heck reaction was used for the industrial synthesis of a number of important products in quantities of more than 1 ton per year. Among them, the herbicide is prosulfuron , obtained by the reaction of 2-sulfonatobenzene diazonium with 3,3,3-trifluoropropene, and a sunscreen 2-ethylhexyl- n- methoxycinnamate. Naproxen , an anti-inflammatory drug, is prepared by Heck reaction between 2-bromo-6-methoxynaphthalene and ethylene and the subsequent carbonylation reaction. The Heck reaction is compatible with many functional groups, which allows it to be used in the final stages of the synthesis of complex compounds. So, it was a key stage in the synthesis of the anti-asthma drug singular. The mild reaction conditions allowed it to be used for combining unstable aromatic bromides to obtain monomers of coatings for electronics components [22] .
Notes
- ↑ 1 2 3 4 CONRR, 2010 .
- Smith, Dilman, 2009 , p. 111.
- Smith, Dilman, 2009 , p. 112
- 2 1 2 3 Smith, Dilman, 2009 , p. 112-113.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3011-3012.
- ↑ 1 2 Beletskaya, Cheprakov, 2000 , p. 3012.
- ↑ 1 2 de Meijere, Meyer, 1995 , p. 2381.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3015.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3017.
- Smith, Dilman, 2009 , p. 113.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3050-3052.
- ↑ 1 2 Beletskaya, Cheprakov, 2000 , p. 3052-3053.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3053-3054.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3020.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3024-3025.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3029-3030.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3046-3050.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3054-3055.
- ↑ 1 2 Beletskaya, Cheprakov, 2000 , p. 3030-3032.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3038.
- ↑ Beletskaya, Cheprakov, 2000 , p. 3041-3042.
- V de Vries JG The Heck reaction in the production of fine chemicals (Eng.) // Can. J. Chem. - 2001. - Vol. 79 . - P. 1086-1092 . - DOI : 10.1139 / cjc-79-5 / 6-1086 .
Literature
- Russian-speaking sources
- Smith, V.A., Dilman, A.D. Chapter 5.3. Heck's reaction // Basics of modern organic synthesis. - M .: Bean. Laboratory of Knowledge, 2009. - P. 111-117. - ISBN 978-5-94774-941-0 .
- Review articles
- Beletskaya IP, Cheprakov AV, The Heck Reaction, as a Sharpening Stone of Palladium Catalysis (Eng.), Chem. Rev. - 2000. - Vol. 100 , no. 8 - P. 3009-3066 . - DOI : 10.1021 / cr9903048 . - PMID 11749313 . Archived August 19, 2013.
- de Meijere A., Meyer's Feathers Feathers: Make It Your Own Peace: The Heck Reaction in Modern Garb (Eng.) // Angew. Chem. Int. Ed. - 1995. - Vol. 33 , no. 23-24 . - P. 2379-2411 . - DOI : 10.1002 / anie.199423791 .
- Heck Reaction // Comprehensive Organic Name Reactions and Reagents. - Wiley, 2010. - DOI : 10.1002 / 9780470638859.conrr301 .
- Original work
- Heck RF Acylation, methylation, and carboxyalkylation of olefins by Group VIII metal derivatives (Eng.) // J. Am. Chem. Soc. - 1968. - Vol. 90 , no. 20 - P. 5518-5526 . - DOI : 10.1021 / ja01022a034 .
- Heck RF Mechanism of arylation and carbomethoxylation of olefins with organopalladium compounds (Eng.) // J. Am. Chem. Soc. - 1969. - Vol. 91 , no. 24 - P. 6707-6714 . - DOI : 10.1021 / ja01052a029 .
- Mizoroki T., Mori K., Ozaki A. Arylation of Olefin with Aryl Iodide Catalyzed by Palladium (English) // Bull. Chem. Soc. Jap .. - 1971. - Vol. 44 . - P. 581 . - DOI : 10.1246 / bcsj.44.581 .
- Heck RF, Nolley Jr. JP Palladium-catalyzed vinylic hydrogen substitution reactions with aryl, benzyl, and styryl halides (English) // J. Org. Chem. - 1972. - Vol. 37 , no. 14 - P. 2320-2322 . - DOI : 10.1021 / jo00979a024 .