Transferrin is a blood plasma protein that transports iron ions . Transferrin is a glycosylated protein that binds iron ions firmly but reversibly. About 0.1% of all iron ions in the body are associated with transferrin (which is about 4 mg), however, iron ions associated with transferrin are of great importance for metabolism . Transferrin has a molecular weight of about 80 kDa and has two Fe 3+ binding sites. The affinity of transferrin is very high (10 23 M −1 at pH 7.4), but it progressively decreases with decreasing pH below the neutral point. When transferrin is not bound to iron, it is an apoprotein .
| Transferrin | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | |||||||||||||
| |||||||||||||
| Identifiers | |||||||||||||
| , PRO1557, PRO2086, TFQTL1, HEL-S-71p, transferrin | |||||||||||||
| External IDs | |||||||||||||
| |||||||||||||
| RNA expression profile | |||||||||||||
  | |||||||||||||
| Ortologists | |||||||||||||
| Kinds | Person | Mouse | |||||||||||
| Entrez | |||||||||||||
| Ensembl | |||||||||||||
| UniProt | |||||||||||||
| RefSeq (mRNA) | |||||||||||||
| RefSeq (protein) | |||||||||||||
| Locus (UCSC) | |||||||||||||
| PubMed Search | |||||||||||||
| Wikidans | |||||||||||||
| View / Edit (Man) | View / Edit (Mouse) | ||||||||||||
Transport Mechanism
When transferrin is bound to iron ions, a transferrin receptor on the cell surface (for example, red blood cell precursors in the red bone marrow) joins it and, as a result, enters the cell in the vesicle. Then the pH inside the bubble goes down due to the work of the proton ion pumps , causing these transferrin to release iron ions. The receptor moves back to the surface of the cell, ready to bind transferrin again. Each transferrin molecule can transfer 2 iron ions Fe 3+ at once.
The gene encoding transferrin in humans is located in the region of the third chromosome 3q21. Studies conducted on royal serpents in 1981 showed that transferrin is inherited by a codominant mechanism .
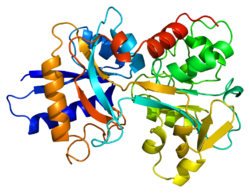
Structure
In humans, transferrin is a polypeptide chain consisting of 679 amino acids . This is a complex consisting of alpha-helices and beta-layers, which form 2 domains (the first is located at the N-end, and the second at the C-end). N- and C- terminal sequences are represented by spherical lobes, between which there is an iron binding site. The amino acids that bind iron ions to transferrin are identical for both fractions: 2 tyrosine , 1 histidine , 1 aspartic acid . To bind an iron ion, an anion is required, preferably a carbonate ion (CO 3 2− ). Transferrin also has a transferrin receptor: it is a disulfide-linked homodimer . [1] In humans, each monomer consists of 760 amino acids. Each monomer consists of 3 domains : an apical domain, a spiral domain, a protease domain.
Tissue distribution
The liver is the main source of transferrin production, but other tissues, such as the brain , also produce these molecules. The main role of transferrin is the delivery of iron from the absorption centers in the duodenum and the digestion of erythrocytes by macrophages to all tissues. Transferrin plays an especially important role in active cell division, for example, in hematopoiesis . The transferrin itself contains the protein itself called transferrin , as well as ovotransferrin , lactoferrin , melanotransferrin , carbonic anhydrase inhibitor , saxifilin , the main protein of sea urchins , crayfish protein , pacfastin , green algae protein . [2]
Immune system
Transferrin take part in ensuring innate immunity. Transferrins are present in the mucous membranes, where they bind iron ions. As a result of reducing the concentration of free iron ions, only a small fraction of the bacteria is able to multiply under such conditions.
Indicators of the norm (g / l) in the analysis for transferrin may be different for children and adults, men and women (for women it is slightly higher), but in general the content of transferrin should be in the range from 1.3 to 3.8.
The concentration of transferrin is reduced in inflammatory processes, [3] , nephrotic syndrome, liver cirrhosis, hemochromatosis, and some other conditions.
In most cases, iron deficiency anemia is accompanied by an increase in transferrin levels. [1] In some cases (congenital antransferrinemia , the presence of antibodies to transferrin, a decrease in transferrin due to a general protein deficiency), iron deficiency anemia, on the contrary, may be due to a violation of iron transport due to a reduced level of transferrin [4] .
Other effects
The metal binding properties of transferrin greatly affect the biochemistry of plutonium in humans. . Transferrin has a bactericidal effect, as it makes Fe 3+ inaccessible to bacteria. Local hemosiderosis is capable of increasing the overall level of transferrin, which may explain many of the effects of autohemotherapy.
Ethnic polymorphism
Some variants of transferrin are more common in populations of a specific anthropological group. So, TF * DChi is a marker of Mongoloid populations and it is widely distributed in East Asia [5] . Variants that differ from the most common type of TF CC were rarely encountered, therefore the information content of this system from the population-genetic position was small. With the introduction of the isoelectric focusing method into practice, genetic heterogeneity of variant C was established [6] . The two most common alleles of TF * C1 and TF * C2 are present in all populations. In Europeans and American white TF * C1 occurs with a frequency of 75-78%. TF * C2 among them varies with frequencies of 13-19%, the limits of variation in Asian populations range from 15 to 34%. Estimation of the frequency of TF * C3 seems to be useful in population genetic studies. This allele occurs with a frequency of 4–7% in Caucasoid groups , with a frequency of 1–4% in the populations of India , sporadic cases of occurrence of this factor in East Asia, some groups of the Pacific Basin and in American Negroes. In other populations, it is completely absent. According to some authors, the C3 allele, apparently, has a European origin [7] . There are more than 10 subtypes TF * C [6] .
Notes
- 2 1 2 Macedo MF, de Sousa M. Transferrin and the transferrin receptor - 2008. - March ( vol. 7 , no. 1 ). - P. 41-52 . - PMID 18473900 . Archived January 18, 2017.
- Amber Lambert LA, Perri H., Halbrooks PJ, Mason AB, and Comp. Biochem. Physiol. B, Biochem. Mol. Biol. : journal. - 2005. - October ( vol. 142 , no. 2 ). - P. 129-1141 . - DOI : 10.1016 / j.cbpb.2005.07.007 . - PMID 16111909 .
- ↑ Ritchie RF, Palomaki GE, Neveux LM, Navolotskaia O., Ledue TB, Craig WY; .) // J. Clin. Lab. Anal. : journal. - 1999. - Vol. 13 , no. 6 - P. 273-279 . - DOI : 10.1002 / (SICI) 1098-2825 (1999) 13: 6 <273 :: AID-JCLA4> 3.0.CO; 2-X . - PMID 10633294 .
- ↑ A.L.Tikhomirov, S.I. Sarsania, A.A. Kocharyan - Iron deficiency anemia: actual problem, adequate treatment. (inaccessible link) . The appeal date is November 28, 2012. Archived January 18, 2013.
- ↑ Kamboh MI, Ferrell RE Human transferrin polymorphism (Eng.) // Hum. Hered. - 1987. - Vol. 37 . - P. 65-81 .
- ↑ 1 2 Prokop O., Geler V. Groups of human blood. - Moscow: Medicine, 1991. - 512 p.
- ↑ Sikstrom C., Nylander PO Transferrin subtypes and ethnic heterogeneity in Sweden (Eng.) // Hum. Hered. - 1990. - Vol. 40 - P. 335—339 .